Trans-Masculine Hormone Replacement Therapy
Time to read 15 mins
Introduction
Within this section of text, cross sex hormones for feminine to trans masculine and trans nonbinary individuals will be discussed. Medication’s will be divided into forms of testosterone and progestins used to aid transition in certain cases.
The aim of hormone therapy in FtM individuals is reducing the effects of estrogen and inducing masculine secondary sex characteristics. This can include but is not limited to →
Additionally, skin may become tougher and breast tissue may lose glands to produce milk. An in-depth chart of all effects and timelines can be found under the testosterone gel subsection (Bourns et al., 2023 p. 65).
Changes such as the deepening of voice and clitoral growth are irreversible. Other changes, such as fat redistribution and increased muscle mass, are generally reversible. The menstrual cycle will generally stop within 3–6 months of taking testosterone, but this does not eliminate fertility, it just decreases the chance (Bourns et al., 2023 p. 65).
The effects of testosterone vary based on age, genetics, ethnicity, body habits, lifestyle, and dose. The first changes are thickening of body hair and deepening of voice, both of which take years to mature. Similarly, clitoral growth begins in the first few months and can be accompanied by mild clitoral discomfort and increased spontaneous arousal (Bourns et al., 2023 p. 65).
The main ways to administer testosterone are via injection or gel. Testosterone patches were stopped in 2021 (Bourns et al., 2023 p. 1.3; p. 66).
For patients taking a low dose of testosterone, or taking it trans dermally (through the skin), a progestin may be introduced. Options include →
These will be discussed in more depth later. Reversibility of changes is contingent on time spent on hormones along with what the desired change looking to be reversed is. Some changes are permanent.
Please consult a general practitioner if you wish to undergo hormone replacement therapy.
Effects
- Stopping menstruation
- Clitoral growth
- Increased muscle mass
- Fat redistribution
- Body hair growth, including facial hair
- Deepened voice
- Levonorgestrel-releasing intra-uterine system (IUS) (e.g., “Mirena”)
- Injectable medroxyprogesterone acetate (MPA) (“Depo-Provera”)
(Bourns et al., 2023 p. 65)
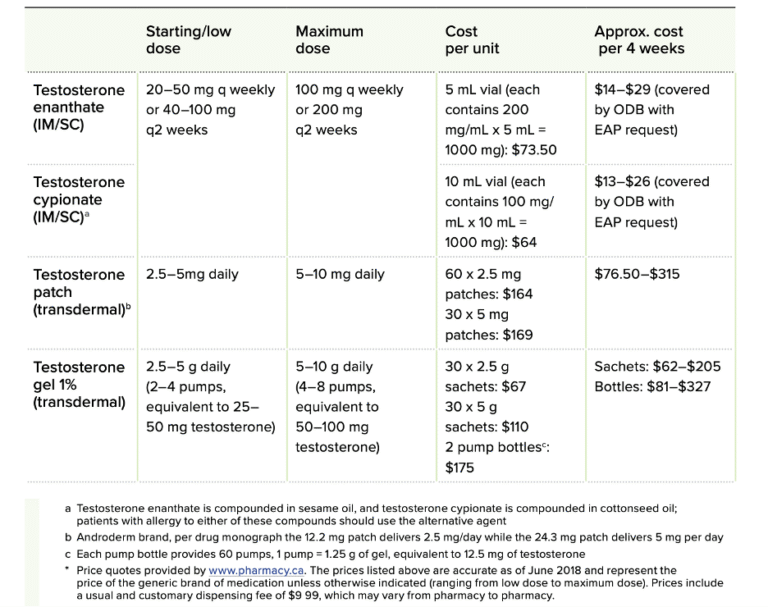
Types of Injectable Testosterone
Testosterone Enanthate can be administered either intramuscularly or subcutaneously (under the skin).
It is covered by the Ontario Drug Benefits (ODB) program with an Exceptional Access Program request (EAP). With an approved EAP, the cost should be $14–29 per vial containing 1000 mg of medicine. Without an EAP, the cost is $73.50 per vial (Bourns et al., 2023 p. 70).
Testosterone Cypionate is the other common type of injectable testosterone and can also be injected intramuscularly or subcutaneously.
The only difference between the two medications is that testosterone Enanthate is compounded in sesame oil, and testosterone Cypionate is compounded in cottonseed oil. This means patients with an allergy to one should be able to take the other (Bourns et al., 2023 p. 70).
Testosterone Cypionate is also covered by Ontario Drug Benefits with an Exceptional Access Program request. With coverage, it costs $13–26 per vial of 1000 mg of medicine, or $64 without coverage (Bourns et al., 2023 p. 70).
Dosage
- Typical starting dose: 20–50 mg weekly or 40–100 mg bi-weekly
- Maximum dose: 100 mg weekly or 200 mg bi-weekly
Risks and Considerations
Data and studies regarding the risk of taking testosterone over an extended period of time have reached different conclusions. Much of this research focuses on cisgender men, who are not as at risk for cardiovascular disease (CV).
Studies specifically on trans-masc individuals have come to different conclusions regarding whether complications due to cardiovascular disease were increased and, if so, by how much (Bourns et al., 2023 p. 76).
The general consensus is that there is an increase in CV risk, and that increase is statistically significant. However, unless you have pre-existing risk factors such as PCOS, the addition of testosterone will not lead to significant danger. Patients with PCOS should consult their doctor, as the risk will be magnified by both and should be carefully considered (Bourns et al., 2023 p. 76).
Patients with PCOS or other androgen disorders should also be cautious of testosterone converting into estrogen, which can lead to an increased risk of endometrial cancer. If unexplained, frequent, irregular, or heavy bleeding occurs when starting testosterone, consult your doctor immediately (Bourns et al., 2023 p. 78).
Finally, pelvic pain is somewhat common within the first 6 months. Diagnoses vary greatly and can include vaginitis, cystitis, STIs, post-surgical sequelae, as well as musculoskeletal and psychological causes. If you are experiencing abnormal pelvic pain unrelated to cramps, consult your doctor (Bourns et al., 2023 p. 79).
Other Considerations
- Sleep apnea worsening or beginning
- Increased hemoglobin or red blood cells (not a concern unless above the typical male range)
- Fluctuations in energy and mood, often dependent on time since last injection
- Reports of decreased energy as more time passes after a shot
- Increased aggression, hypersexuality, and rare psychotic symptoms
(Bourns et al., 2023 p. 77)
Gel Based Testosterone
Testosterone-based gel shares many similarities with testosterone-based injections. This section is dedicated to the differences found, including effectiveness, differing side effects, and pros and cons.
The main benefit of testosterone gel is the consistency that comes with daily application. Patients taking testosterone gel almost never experience the same degree of fatigue or moodiness that can occur with injections (Bourns et al., 2023 p. 67).
The trade-offs can feel significant by comparison. Testosterone gel should be applied to the upper arms, and a two-hour window is necessary for the gel to dry. →
Some patients also report that testosterone gel has an unpleasant odor (Bourns et al., 2023 p. 67).
A typical starting dose is 2.5–5 g daily, which can be increased to a maximum dose of 10 g per day.
Additional Info
During this two-hour window:
- The skin should not touch clothing that could absorb the gel
- The skin should not come into contact with other people, as they may be exposed to excessive amounts of testosterone
Testosterone gel is not covered by Ontario Drug Benefits. Costs include:
1. $110 for 30 sachets, each containing 5 g of gel
2. $175 for two pump bottles with 60 pumps each
Four pumps are equivalent to 5 g of testosterone, or an average daily dose (Bourns et al., 2023 p. 70).
At a dose of 5 g per day:
1. Sachets cost approximately $133 per month
2. Pump bottles cost approximately $204 per month
Progestins and gnRH
Progestins are generally not part of hormone therapy in trans-masc or trans-nonbinary patients. That said, there are situations where their use can be warranted.
Approximately 1 out of 10 transmasculine people do not have their periods suppressed on testosterone alone. This is particularly common at lower doses (Dimakopoulou & Seal, 2024).
To address this, progestins may be used, including →
Levonorgestrel can also be used without testosterone to prevent periods (Bourns et al., 2023 p. 65).
Lupron (GnRH)
Another option is Lupron, an injectable gonadotropin-releasing hormone (GnRH). Normally, the brain releases GnRH to signal the pituitary gland to produce hormones.
Lupron floods these receptors, causing a brief increase in hormone production before the pituitary gland becomes desensitized and stops sending signals entirely, effectively halting hormone production (Bourns et al., 2023 p. 41).
Possible side effects of Lupron include →
(BC Cancer Agency, 2020 p. 4)
Lupron dosing options include →
(BC Cancer Agency, 2020 p. 7)
The injection is intramuscular and is typically administered in the thigh or buttock. Muscles may tense and cramp for several hours following the injection.
Lupron is generally not covered by the Ontario Drug Benefit program. Costs are approximately →
(Government of Ontario, Ministry of Health, n.d.)
There are some ways to obtain coverage, but this requires a doctor to state that Lupron is medically necessary as an anti-androgen.
Additional Info
- Medroxyprogesterone acetate at 10 mg per dose, taken three times a day
- Levonorgestrel-releasing IUS, which is inserted into the uterus and slowly releases levonorgestrel to suppress menstruation
(Dimakopoulou & Seal, 2024; Bourns et al., 2023 p. 65)
- Anemia
- Fatigue
- Weight gain
- Hot flashes
- Decreased bone mineral density
- Loss of muscle mass
- Depression
- Memory loss
- 7.5 mg injected once monthly
- 22.5 mg injected once every three months
- $388 for a 7.5 mg injection
- $1159 for a 22.5 mg injection
References
BC Cancer Agency. (2020, April 1). Leuprolide monograph [PDF]. BC Cancer. https://www.bccancer.bc.ca/drug-database-site/Drug%20Index/Leuprolide_monograph.pdf
Bourns, A., Edgar, A., Lou, A., Kucharski, E., Townsend, M., Fung, R., Hranilovic, S., Weisdorf, T., & Rainbow Health Ontario. (2023). Guidelines for gender-affirming primary care with trans and non-binary patients (4th ed., with 2023 updates) [PDF]. Sherbourne Health / Rainbow Health Ontario. https://www.rainbowhealthontario.ca/wp-content/uploads/2021/09/Guidelines-FINAL-4TH-EDITION-With-2023-Updates.pdf
Deutsch, M. B. (2016, June 17). Overview of masculinizing hormone therapy. UCSF Gender Affirming Health Program. Retrieved October 10, 2025, from https://transcare.ucsf.edu/guidelines/masculinizing-therapy
Dimakopoulou, A., & Seal, L. J. (2024). Testosterone and other treatments for transgender males and non-binary trans masculine individuals. Best Practice & Research Clinical Endocrinology & Metabolism, 38(5), Article 101908. https://doi.org/10.1016/j.beem.2024.101908
Government of Ontario, Ministry of Health. (n.d.). Lupron Depot PDS (leuprolide acetate) — Formulary entry. Ontario Drug Benefit Formulary. Retrieved [date you accessed it], from https://www.formulary.health.gov.on.ca/formulary/results.xhtml?q=%22Lupron+Depot+PDS%22&type=2
